You should upgrade or use an alternative browser.
Synthesis and Pyrolysis of UR-144: A Detailed Guide
Synthesis and Pyrolysis of UR-144: A Detailed Guide
UR-144 cannabinoid synthesis is represented in this topic.
Laboratory requirements:
The working place (a tabletop for laboratory syntheses and manipulations) must be supplied with exhaust ventilation and fresh air inflow. laboratory have to be equipped with multi-tiered racks for product drying. They have to be also supplied with good air ventilation. It is also worth to take care about weighing and packaging place. The place should has enough space, be convenient and be located under exhaust ventilation (preferably). Laboratory floors are recommended to be covered with a polyethylene film during synthesis procedures by reason that cannabinoid dust abundance cannot be avoided. Subsequently, the laboratory space will be more convenient to clean up by collecting polyethylene film.
Reagents:
1. 2,2,2,3,3-tetramethylcyclopropyl(1H-indol-3-yl)methanone (TMCP-indole);
2. Sodium hydroxide (NaOH);
3. Tetra-n-butylammonium bromide (TBAB) CAS 1643-19-2;
4. 1-Bromopentane (1-BP) CAS 110-53-2;
5. Distilled water (H2O);
Equipment and glassware:
1. Laboratory scale (0.1-200 g is suitable);
2. Flat-bottom flasks 10 l;
3. Funnel for bulk materials (with wide bottom);
4. Scoops for flour;
5. Beakers 1-2 l;
6. Electric burner;
7. Overhead stirrer (optional);
8. Silicone baking molds;
9. Syringes;
10. Blender;
11. Large bowl or bucket;
Individual protection:
1. Long sleeve chemical coat;
2. Respirator mask (or respirator + goggles);
3. Latex gloves;
4. Fabric gloves or a piece of thick fabric;
Reaction:
1. TMCP-indol 2000 g is weighted and put into 10 l flask through a funnel for bulk materials;
2. Sodium hydroxide (NaOH) 1300 g is weighted and put into same flask;
3. TBAB 800 g is also weighted and put into same 10 l flask;
4. 1-Bromopentane 1500 g is poured into the flask;
5. Fresh water 3000 ml is poured there (better to use distilled water);
The reaction mixture is stirred after reagents addition until NaOH is dissolved completely and two layers are formed. You can use cotton gloves under latex one to avoid burns, which can happen because NaOH dissolving is conducted with heat releasing. Also, it is recommended to use retort clamp apparatus.
As soon as two layers are divided and the all sodium hydroxide are dissolved, the flask is put onto the heating plate and the heating is turned on to the maximum power. Top stirrer is installed and turned on. In case you don't have a top stirrer, you have to use heat resistant gloves to avoid burns. The reaction mixture have to be stirred well in order to mix layers (be homogenized).
Top stirrer have to work during whole reaction procedure. In case you don't have top stirrer, reaction flask is shakes vigorously during whole reaction procedure. The simmer of reaction mixture is supplied until the mixture is become milky and without separated layers (homogeneous). It takes 15-30 min usually.
Next, heating plate is removed and reaction mixture is left for layer separation. Top layer is needed. Product can crystalize directly in the reaction flask, which makes it difficult to extract. According this fact, you have to do separation procedure quickly.
Top layer is moved into a beaker or directly into silicone molds (do not forget about the temperature, use thick gloves or towels). Top layer (oil) can be collected by a syringe. When the layer is become very thin, mixture can be poured into a measuring cylinder for much convenient handling.
Silicone molds with an oil layer are left for 24 hours to solidification. Silicone molds are used for more convenient handling because oil in these molds are solidified and taken their form.
Crystallized product is taken out and crushed. Than, UR-144 is placed onto trays, dried in well ventilated dry warm room. Usually, it takes 2 days. Also, you can use vacuum desiccator to dry it much faster. The yield is 3000 g.
This reaction procedure can be used for other JWH row cannabinoids.
 UR-144 Pyrolysis Products
UR-144 Pyrolysis Products
UR-144 (TMCP-018, KM-X1, MN-001, YX-17; CAS number 1199943-44-6) contains a cyclopropane ring, which is thermally unstable, and this may be of concern as the product is normally consumed by smoking. Cyclopropane itself is susceptible to thermal isomerization to propylene at the temperatures approximately above 420 °C. Other pyrolysis products have been reported, although they formed at much higher temperatures. In general, both substituted and unsubstituted cyclopropanes undergo a variety of ring-opening reactions. As the temperature of the tip of a burning cigarette can exceed 700 °C, this would suggest that cyclopropane moiety in UR-144 may undergo some of these transformations. In the work presented here, a number of herbal, resin-type products and powders purchased from online vendors were analyzed. UR-144 was identified in the products along several related compounds, one being a novel hydrated derivative. The pyrolysis of UR-144 was also studied. In this article, you can read about heating process and thermal decomposition of substances while smoking. There is UR-144 synthesis method from TMCP-indole.
The synthetic cannabinoid, UR-144 ((1-pentyl-1H-indol-3-yl)(2,2,3,3-tetramethylcyclopropyl)methanone), was identified in commercial ‘legal high’ products (herbal, resin, and powder). Along with this, six related compounds were detected. The most abundant one (2.1) was identified as 4-hydroxy-3,3,4-trimethyl-1-(1-pentyl-1H-indol-3-yl)pentan-1-one, a product of the electrophilic addition of water to the cyclopropane moiety in UR-144. Compound 2.1 was found to be undergoing cyclization which leads to the formation of two additional interconvertible compounds (2.3, tentatively identified as 1-pentyl-3-(4,4,5,5-tetramethyl-4,5-dihydrofuran-2-yl)-1H-indole which is stable only in absence of water and also observed as GC artifact) and 2.2, a protonated derivative of 2.3 which is formed in acidic solutions. The remaining compounds were identified as possible degradation products of the group 2 compounds (4,4,5,5-tetramethyldihydrofuran-2(3H)-one and 1-pentylindoline-2,3-dione) and intermediates or by-products from the synthesis of UR-144 ((1H-indol-3-yl)(2,2,3,3-tetramethylcyclopropyl)methanone, 1-pentyl-1H-indole and 1-(1-pentyl-1H-indol-3-yl)hexan-1-one). Pyrolysis of herbal products containing the group 2 compounds or UR-144 resulted in the formation of 3,3,4-trimethyl-1-(1-pentyl-1H-indol-3-yl)pent-4-en-1-one (3). This was confirmed by separate pyrolysis of 2.1 and UR-144. Also, the two additional minor compounds, 1-(1-pentyl-1H-indol-3-yl)ethanone and 1-(1-pentyl-1H-indol-3-yl)propan-1-one, were detected. Pathways for these transformations are presented.
Discussion
Synthetic cannabinoids have become one of the primary challenges of forensic toxicology and seized drug analysis since their appearance in the mid-2000s. Although the synthetic cannabinoids act on the cannabinoid receptors, the pharmacological effects are markedly different from that of marijuana, which suggests mechanisms of toxicity separate from any caused through binding interactions. It has been noted that pyrolysis products may contribute to the observed psychological effects.
Fig.1 Reported pyrolytic products of two synthetic cannabinoids.
Upper frame: UR-144 forming 3,3,4-trimethyl-1-(1-pentyl-1H-indol-3-yl) pent-4-en-1-one;
Lower frame: XLR11 forming 1-(1-(5-luoropentyl)-1H-indol-3-yl)-3,3,4-trimethylpent-4-en-1-one. In both cases, the cyclopropane ring is broken to create an isobutylene group.
UR-144 (33, Fig.1), ((1-pentyl-1H-indol-3-yl) (2,2,3,3-tetramethylcyclopropyl)methanone), is an indole based synthetic cannabinoid which is structurally similar to JWH-018 in that both molecules contain a pentyl side chain off the nitrogen of the in-dole core and a secondary ring structure bridged to the indole via a carbonyl group. This secondary ring structure is the only difference between these two cannabinoids, with the naphthalene substituent of JWH-018 being replaced with a tetramethylcyclopropane group in UR-144. The tetramethylcyclopropane moiety, like other cyclopropane derivatives, is considered thermally unstable. In 2012, an article was published, which tentatively characterized the main pyrolytic product of UR-144 as 3,3,4-trimethyl-1-(1-pentyl-1H-indol-3-yl)pent-4-en-1-one (34, Fig.1). Several commercially available products containing UR-144 were purchased and analyzed with GC-MS and LC-MS/MS. To simulate the burning process, pyrolysis of two herbal samples was carried out in a quartz tube. While this main pyrolytic product (Structure 34) was observed in the pyrolyzed samples using both detection methods, it was also observed in samples that were not pyrolyzed but analyzed using GC-MS. This finding suggests that the compound is thermally unstable and will be observed as an artifact as a result of volatilization in gas chromatography. Other minor products were characterized by the authors in both extracted samples and the two pyrolyzed samples. Quantitative results were not provided, and the identification of the main pyrolysis product was not confirmed using reference standards. A follow-up study evaluated urine samples for the presence of the main pyrolytic structure (34) as well as numerous metabolites of UR-144.
Fig. 2 Proposed scheme of reactions for compounds related to UR-144.
A number of studies have indicated that most aminoalkylindoles based synthetic cannabinoids undergo extensive metabolism and are often times not detectable in human urine samples. Mono-hydroxylation, di-hydroxylation, carboxylation, and dealkylation metabolites of the major pyrolytic product were tentatively identified. Of the 37 metabolites tentatively identified in this article, 21 were reported products of the main pyrolytic product and thus may have utility as biomarkers of smoked UR-144 in screening assays. Quantitative results were not provided, and the identification of the main pyrolytic structure (34) was not confirmed. A 2013 case study on samples collected from an individual under the influence of UR-144 reported the presence of the parent drug and the pyrolytic product (34) in blood, as well as the respective metabolites in urine. Both UR-144 and its main pyrolytic product (structure 34) were also observed upon analysis of powder residue found in a plastic bag confiscated from the intoxicated individual. Finally, a method validation study was published by Amaratunga et al. in 2014 in which a method was developed to detect XLR11 (35, Fig.1) ((1-(5-fluoropentyl)-1H-indol-3-yl)(2,2,3,3-tetramethylcyclopropyl)-methanone) and UR-144 parent, metabolites, and pyrolysis products in oral fluid. The main pyrolytic product of XLR11 (1-(1-(5-fluoropentyl)-(1H-indol-3-yl)-3,3,4-trimethylpent-4-en-1-one)) was (36, Fig.1) was confirmed using reference standards.
Conclusion
UR-144 was found to be relatively unstable, and facile cyclopropane ring fusion was noted. The thermal isomerization of UR-144 without air (i.e. GC injector port/column) or in the presence of air (i.e. burning of UR-144 containing products) leads to opening of cyclopropane ring and formation of a trimethylbutene product (3). Hydration of the cyclopropane in UR-144 or the trimethylbutene moiety in compound 3 results in the formation of a group of interconvertible compounds. The pharmacological properties of these compounds are unknown, and they can contribute to intensive psychological effects noted following the consumption of UR-144. These compounds may be of interest as individual novel synthetic cannabinoids for use.
-

Highest quality Cannabinoid Precursor (5FADB/5CLADB/ADBB/FUB/JWH/Sgt etc. )and Raw materials supply, 100% Technical Support, 100% Quality Assurance.
FLYRCLAB: We don't just create the present, we help you create the future! At FLYRCLAB, we specialize in the production of high quality pharmaceutical intermediates (precursors). Our quest for unrivaled quality and purity has always been our commitment. Our products undergo rigorous quality...- flyrc
- Thread
- #5cladb #cannabinoids 5f-sgt-151 5fadb ab-chminaca adb butinaca adb-pinaca jwh 018 mdmb-4en-pinaca ur-144
- Replies: 129
- Forum: Sellers of Chemicals and Equipment
-
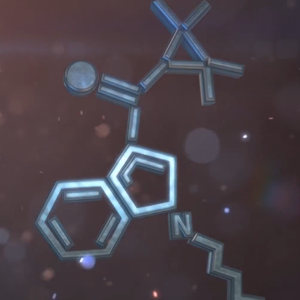
UR-144 synthesis from TMCP-indole (large scale). Video manual.
https://bbgate.com/threads/synthesis-of-ur-144-from-tmcp-indole-large-scale.199/- HEISENBERG
- Media item
- cannabinoid synthesis video cas 1199943-44-6 cas 895152-66-6 km-x1 tmcp-018 tmcp-indole ur-144 ur-144 synthesis ur-144 video
- Comments: 5
- Category: Synthesis of Cannabinoids
-

UR-144 pyrolysis products
Introduction UR-144 (TMCP-018, KM-X1, MN-001, YX-17; CAS number 1199943-44-6) contains a cyclopropane ring, which is thermally unstable, and this may be of concern as the product is normally consumed by smoking. Cyclopropane itself is susceptible to thermal isomerization to propylene at the...- G.Patton
- Thread
- km-x1 mn-001 pyrolysis tmcp-018 ur-144 yx-17
- Replies: 4
- Forum: Cannabinoids
-

TMCP-indole synthesis (UR-144 intermediate, cas 895152-66-6). Large scale.
Reaction scheme: Equipment and glassware: 50 L Batch reactor, which is equipped with drip funnel, top stirrer, thermometer, temperature control system (cooling) and reflux condenser; Funnel; Several buckets 10 and 20 L; Vacuum source; Laboratory scale (1-2000 g is suitable); Measuring cylinder...- WillD
- Thread
- a-834735 cannabinoid synthesis cas 24303-61-5 cas 895152-66-6 indole tmcp-indole synthesis ur-144 xlr-11
- Replies: 15
- Forum: Cannabinoids
-

UR-144 synthesis from TMCP-indole (large scale). Complete video tutorial.
UR-144 cannabinoid synthesis is represented in this topic. Laboratory requirements: The working place (a tabletop for laboratory syntheses and manipulations) must be supplied with exhaust ventilation and fresh air inflow. laboratory have to be equipped with multi-tiered racks for product...- WillD
- Thread
- cannabinoid synthesis video cas 1199943-44-6 cas 895152-66-6 km-x1 tmcp-018 tmcp-indole ur-144 ur-144 synthesis ur-144 video
- Replies: 54
- Forum: Cannabinoids
About Us
Our team brings together the best specialists from different fields.
We are ready to share our experience, discuss difficult issues and find new solutions.
Mirror Links
Help Pages
Follow us
Popular Tags
-
Free product samples
Testing products from new vendors and manufacturers.
Get free samples for testing now!
-
Always stay in touch with BB forum. Element/Matrix.
Connect notifications to always stay in touch with the forum!
Connect
